What Would Happen to the Equilibrium Expressed by the Reaction in Equation 11.8
11.eight: Le Châtelier's Principle- How a System at Equilibrium Responds to Disturbances
- Page ID
- 329816
Learning Objectives
- Describe the ways in which an equilibrium system can be stressed
- Predict the response of a stressed equilibrium using Le Chatelier's principle
Every bit we saw in the previous section, reactions proceed in both directions (reactants get to products and products go to reactants). We tin can tell a reaction is at equilibrium if the reaction quotient (\(Q\)) is equal to the equilibrium abiding (\(K\)). We next accost what happens when a organization at equilibrium is disturbed then that \(Q\) is no longer equal to \(One thousand\). If a system at equilibrium is subjected to a perturbance or stress (such as a change in concentration) the position of equilibrium changes. Since this stress affects the concentrations of the reactants and the products, the value of \(Q\) volition no longer equal the value of \(K\). To re-establish equilibrium, the system will either shift toward the products (if \((Q \leq K)\) or the reactants (if \((Q \geq Chiliad)\) until \(Q\) returns to the same value as \(Yard\). This process is described past Le Chatelier's principle.
Le Chatelier's principle
When a chemic system at equilibrium is disturbed, information technology returns to equilibrium by counteracting the disturbance. As described in the previous paragraph, the disturbance causes a change in \(Q\); the reaction will shift to re-institute \(Q = K\).
Predicting the Direction of a Reversible Reaction
Le Chatelier's principle tin be used to predict changes in equilibrium concentrations when a system that is at equilibrium is subjected to a stress. Nonetheless, if we have a mixture of reactants and products that accept not notwithstanding reached equilibrium, the changes necessary to accomplish equilibrium may not be so obvious. In such a case, nosotros can compare the values of \(Q\) and \(K\) for the organisation to predict the changes.
A chemical organisation at equilibrium can be temporarily shifted out of equilibrium past adding or removing one or more of the reactants or products. The concentrations of both reactants and products and then undergo additional changes to return the system to equilibrium.
The stress on the system in Figure \(\PageIndex{i}\) is the reduction of the equilibrium concentration of SCN− (lowering the concentration of one of the reactants would cause \(Q\) to be larger than K). As a consequence, Le Chatelier'due south principle leads us to predict that the concentration of Atomic number 26(SCN)2+ should decrease, increasing the concentration of SCN− function way back to its original concentration, and increasing the concentration of Fe3 + to a higher place its initial equilibrium concentration.
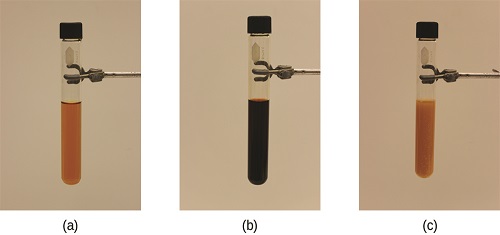
The effect of a change in concentration on a organization at equilibrium is illustrated further by the equilibrium of this chemical reaction:
\[\ce{H2(grand) + I2(g) \rightleftharpoons 2HI(g)} \label{15.7.1a}\]
\[K_c=\mathrm{50.0 \; at\; 400°C} \label{15.vii.1b}\]
The numeric values for this example accept been determined experimentally. A mixture of gases at 400 °C with \(\mathrm{[H_2] = [I_2]} = 0.221\; Grand\) and \(\ce{[HI]} = 1.563 \;G\) is at equilibrium; for this mixture, \(Q_c = K_c = 50.0\). If \(\ce{H_2}\) is introduced into the system so quickly that its concentration doubles before it begins to react (new \(\ce{[H_2]} = 0.442\; M\)), the reaction will shift so that a new equilibrium is reached, at which
- \(\ce{[H_2]} = 0.374\; M\),
- \(\ce{[I_2]} = 0.153\; M\), and
- \(\ce{[Hullo]} = 1.692\; Thou\).
This gives:
\[\begin{align*} Q_c &=\mathrm{\dfrac{[HI]^2}{[H_2][I_2]}} \\[4pt] &=\dfrac{(1.692)^ii}{(0.374)(0.153)} \\[4pt] &= 50.0 =K_c \label{fifteen.seven.two} \finish{align*}\]
Nosotros have stressed this system by introducing additional \(\ce{H_2}\). The stress is relieved when the reaction shifts to the right, using up some (only not all) of the excess \(\ce{H_2}\), reducing the corporeality of uncombined \(\ce{I_2}\), and forming additional \(\ce{HI}\). Le Châtelier's Principle (Changing Concentrations): https://youtu.be/o9jP1yF6E1U
A Video Discussing Le Châtelier's Principle (Changing Concentrations): https://youtu.exist/o9jP1yF6E1U
Effect of Modify in Pressure on Equilibrium
Sometimes we tin can change the position of equilibrium by irresolute the pressure of a organization. However, changes in pressure level have a measurable issue only in systems in which gases are involved, and and so only when the chemical reaction produces a change in the full number of gas molecules in the system. An easy style to recognize such a organisation is to look for dissimilar numbers of moles of gas on the reactant and product sides of the equilibrium. While evaluating force per unit area (besides as related factors like volume), it is important to remember that equilibrium constants are defined with regard to concentration (for Kc ) or fractional pressure (for KP ). Some changes to full force per unit area, like adding an inert gas that is not part of the equilibrium, will change the total pressure but not the partial pressures of the gases in the equilibrium abiding expression. Thus, addition of a gas not involved in the equilibrium will not adjy the equilibrium.
As we increment the force per unit area of a gaseous organisation at equilibrium, either past decreasing the volume of the system or past calculation more of one of the components of the equilibrium mixture, we innovate a stress by increasing the fractional pressures of one or more of the components. In accordance with Le Chatelier's principle, a shift in the equilibrium that reduces the total number of molecules per unit of volume volition be favored because this relieves the stress. The reverse reaction would be favored past a decrease in pressure.
Consider what happens when we increment the pressure level on a organization in which \(\ce{NO}\), \(\ce{O_2}\), and \(\ce{NO_2}\) are at equilibrium:
\[\ce{2NO(g) + O2(chiliad) \rightleftharpoons 2NO2(g)} \label{fifteen.7.iii}\]
The formation of boosted amounts of \(\ce{NO2}\) decreases the total number of molecules in the system because each time ii molecules of \(\ce{NO_2}\) form, a full of iii molecules of \(\ce{NO}\) and \(\ce{O_2}\) are consumed. This reduces the total pressure exerted past the system and reduces, but does not completely relieve, the stress of the increased force per unit area. On the other hand, a subtract in the pressure on the system favors decomposition of \(\ce{NO_2}\) into \(\ce{NO}\) and \(\ce{O_2}\), which tends to restore the pressure.
At present consider this reaction:
\[\ce{N2(g) + O2(g) \rightleftharpoons 2NO(g)} \characterization{15.vii.4}\]
Because in that location is no change in the full number of molecules in the system during reaction, a modify in pressure level does not favor either germination or decomposition of gaseous nitrogen monoxide.
Le Châtelier's Principle (Changes in Pressure level or Volume):
https://youtu.be/fHlFTtn2gxU
Effect of Change in Temperature on Equilibrium
Irresolute concentration or pressure perturbs an equilibrium considering the reaction quotient is shifted abroad from the equilibrium value. Changing the temperature of a arrangement at equilibrium has a different effect: A modify in temperature really changes the value of the equilibrium constant. However, nosotros tin qualitatively predict the event of the temperature modify by treating it as a stress on the system and applying Le Chatelier's principle.
When hydrogen reacts with gaseous iodine, oestrus is evolved.
\[\ce{H2(yard) + I2(g) \rightleftharpoons 2HI(g)}\;\;\ ΔH=\mathrm{−9.4\;kJ\;(exothermic)} \label{15.7.v}\]
Considering this reaction is exothermic, we can write it with heat equally a product.
\[\ce{H2(g) + I2(one thousand) \rightleftharpoons 2HI(k)} + \text{oestrus} \characterization{15.seven.6}\]
Increasing the temperature of the reaction increases the internal energy of the arrangement. Thus, increasing the temperature has the result of increasing the amount of one of the products of this reaction. The reaction shifts to the left to salvage the stress, and at that place is an increase in the concentration of H2 and I2 and a reduction in the concentration of HI. Lowering the temperature of this arrangement reduces the amount of energy present, favors the production of rut, and favors the formation of hydrogen iodide.
When we change the temperature of a system at equilibrium, the equilibrium abiding for the reaction changes. Lowering the temperature in the How-do-you-do system increases the equilibrium constant: At the new equilibrium the concentration of HI has increased and the concentrations of H2 and I2 decreased. Raising the temperature decreases the value of the equilibrium constant, from 67.5 at 357 °C to l.0 at 400 °C.
Temperature affects the equilibrium between \(\ce{NO_2}\) and \(\ce{N_2O_4}\) in this reaction
\[\ce{N2O4(thou) \rightleftharpoons 2NO2(yard)}\;\;\; ΔH=\mathrm{57.20\; kJ} \label{15.7.7}\]
The positive ΔH value tells us that the reaction is endothermic and could be written
\[\ce{estrus}+\ce{N_2O4(m) \rightleftharpoons 2NO2(g)} \label{xv.7.8}\]
At higher temperatures, the gas mixture has a deep brown color, indicative of a pregnant corporeality of brown \(\ce{NO_2}\) molecules. If, yet, we put a stress on the system by cooling the mixture (withdrawing free energy), the equilibrium shifts to the left to supply some of the energy lost by cooling. The concentration of colorless \(\ce{N_2O_4}\) increases, and the concentration of brown \(\ce{NO_2}\) decreases, causing the brown color to fade.
The overview of how unlike disturbances affect the reaction equilibrium properties is tabulated in Tabular array \(\PageIndex{i}\).
| Disturbance | Observed Change as Equilibrium is Restored | Direction of Shift | Event on K |
|---|---|---|---|
| reactant added | added reactant is partially consumed | toward products | none |
| product added | added product is partially consumed | toward reactants | none |
| decrease in book/increase in gas pressure level | pressure decreases | toward side with fewer moles of gas | none |
| increment in volume/subtract in gas pressure | pressure increases | toward side with more moles of gas | none |
| temperature increase | oestrus is absorbed | toward products for endothermic, toward reactants for exothermic | changes |
| temperature decrease | oestrus is given off | toward reactants for endothermic, toward products for exothermic | changes |
Example \(\PageIndex{one}\)
Write an equilibrium abiding expression for each reaction and utilise this expression to predict what will happen to the concentration of the substance in bold when the indicated change is fabricated if the system is to maintain equilibrium.
- \(2HgO_{(s)} \rightleftharpoons 2Hg_{(l)} + \mathbf{O}_{ii(g)}\): the corporeality of HgO is doubled.
- \(NH_4HS_{(s)} \rightleftharpoons \mathbf{NH}_{iii(g)} + H_2S_{(g)}\): the concentration of \(H_2S\) is tripled.
- \( \textbf{n-butane}_{(thou)} \rightleftharpoons isobutane_{(g)}\): the concentration of isobutane is halved.
Given: equilibrium systems and changes
Asked for: equilibrium abiding expressions and effects of changes
Strategy:
Write the equilibrium constant expression, remembering that pure liquids and solids do not appear in the expression. From this expression, predict the alter that must occur to maintain equilibrium when the indicated changes are made.
Solution:
Because \(HgO_{(s)}\) and \(Hg_{(l)}\) are pure substances, they do not appear in the equilibrium abiding expression. Thus, for this reaction, \(Thousand = [O_2]\). The equilibrium concentration of \(O_2\) is a constant and does not depend on the amount of \(HgO\) present. Hence adding more \(HgO\) will not bear on the equilibrium concentration of \(O_2\), and so no compensatory alter is necessary.
\(NH_4HS\) does non announced in the equilibrium abiding expression considering it is a solid. Thus \(Thou = [NH_3][H_2S]\), which means that the concentrations of the products are inversely proportional. If calculation \(H_2S\) triples the \(H_2S\) concentration, for case, then the \(NH_3\) concentration must decrease by about a factor of 3 for the organisation to remain at equilibrium so that the product of the concentrations equals \(Grand\).
For this reaction, \(K = \frac{[isobutane]}{[\textit{n-butane}]}\), so halving the concentration of isobutane means that the n-butane concentration must also decrease by about half if the system is to maintain equilibrium.
Practise \(\PageIndex{1}\)
Write an equilibrium abiding expression for each reaction. What must happen to the concentration of the substance in assuming when the indicated change occurs if the arrangement is to maintain equilibrium?
- \(\ce{HBr (thou) + NaH (s) \rightleftharpoons NaBr (due south)} + \mathbf{H_2(g)}\): the concentration of \(\ce{HBr}\) is decreased past a factor of 3.
- \(\ce{6Li (s)} + \mathbf{N_2(yard)} \ce{ \rightleftharpoons 2Li3N(s)}\): the amount of \(\ce{Li}\) is tripled.
- \(\mathbf{SO_2(g)} + \ce{ Cl2(g) \rightleftharpoons SO2Cl2(l)}\): the concentration of \(\ce{Cl2}\) is doubled.
- Answer a
-
\(K = \dfrac{[H_2]}{[HBr]}\); \([H_2]\) must subtract by well-nigh a factor of 3.
- Answer b
-
\(K = \dfrac{1}{[N_2]}\); solid lithium does non appear in the equilibrium constant expression, so no compensatory modify is necessary.
- Answer c
-
\(Thou = \dfrac{ane}{[SO_2][Cl_2]}\); \([SO_2]\) must decrease by about half.
Le Châtelier's Principle (Changes in Temperature):
https://youtu.be/-P5uGuJZ-r8
Catalysts Do Not Touch on Equilibrium
As we learned during our study of kinetics, a catalyst can speed up the charge per unit of a reaction. Though this increment in reaction charge per unit may cause a system to achieve equilibrium more quickly (by speeding upwards the forrad and opposite reactions), a catalyst has no result on the value of an equilibrium constant nor on equilibrium concentrations. The interplay of changes in concentration or pressure, temperature, and the lack of an influence of a goad on a chemical equilibrium is illustrated in the industrial synthesis of ammonia from nitrogen and hydrogen according to the equation
\[\ce{N2(g) + 3H2(m) \rightleftharpoons 2NH3(g)} \label{15.7.9}\]
A large quantity of ammonia is manufactured by this reaction. Each year, ammonia is amongst the meridian 10 chemicals, past mass, manufactured in the globe. Nigh 2 billion pounds are manufactured in the United States each twelvemonth. Ammonia plays a vital role in our global economy. It is used in the product of fertilizers and is, itself, an important fertilizer for the growth of corn, cotton, and other crops. Large quantities of ammonia are converted to nitric acid, which plays an important function in the production of fertilizers, explosives, plastics, dyes, and fibers, and is also used in the steel manufacture.
Fritz Haber
Haber was born in Breslau, Prussia (before long Wroclaw, Poland) in December 1868. He went on to report chemistry and, while at the University of Karlsruhe, he developed what would later be known equally the Haber process: the catalytic formation of ammonia from hydrogen and atmospheric nitrogen under high temperatures and pressures. For this work, Haber was awarded the 1918 Nobel Prize in Chemistry for synthesis of ammonia from its elements (Equation \ref{fifteen.seven.9}). The Haber process was a boon to agriculture, as it allowed the production of fertilizers to no longer exist dependent on mined feed stocks such as sodium nitrate.

Currently, the annual production of synthetic nitrogen fertilizers exceeds 100 million tons and constructed fertilizer production has increased the number of humans that arable land tin support from 1.ix persons per hectare in 1908 to four.3 in 2008. The availability of nitrogen is a strong limiting factor to the growth of plants. Despite accounting for 78% of air, diatomic nitrogen (\(\ce{N_2}\)) is nutritionally unavailable to a majority of plants due the tremendous stability of the nitrogen-nitrogen triple bond. Therefore, the nitrogen must be converted to a more than bioavailable course (this conversion is chosen nitrogen fixation). Legumes achieve this conversion at ambient temperature by exploiting bacteria equipped with suitable enzymes.
In addition to his work in ammonia production, Haber is also remembered by history as 1 of the fathers of chemical warfare. During Earth War I, he played a major function in the development of poisonous gases used for trench warfare. Regarding his part in these developments, Haber said, "During peace fourth dimension a scientist belongs to the Earth, only during war time he belongs to his country."one Haber defended the use of gas warfare against accusations that it was inhumane, saying that death was decease, by whatsoever ways it was inflicted. He stands equally an case of the ethical dilemmas that face scientists in times of state of war and the double-edged nature of the sword of scientific discipline.
Similar Haber, the products made from ammonia can exist multifaceted. In addition to their value for agriculture, nitrogen compounds tin can also exist used to reach destructive ends. Ammonium nitrate has as well been used in explosives, including improvised explosive devices. Ammonium nitrate was one of the components of the bomb used in the attack on the Alfred P. Murrah Federal Building in downtown Oklahoma City on April 19, 1995.
Summary
Systems at equilibrium tin exist disturbed by changes to temperature, concentration, and, in some cases, volume and pressure; book and pressure level changes will disturb equilibrium if the number of moles of gas is different on the reactant and production sides of the reaction. The system's response to these disturbances is described by Le Chatelier's principle: The organization will respond in a way that counteracts the disturbance. Non all changes to the system result in a disturbance of the equilibrium. Adding a catalyst affects the rates of the reactions simply does not alter the equilibrium, and changing pressure or volume will not significantly disturb systems with no gases or with equal numbers of moles of gas on the reactant and product side.
- 1 Herrlich, P. "The Responsibility of the Scientist: What Can History Teach Us Almost How Scientists Should Handle Research That Has the Potential to Create Harm?" EMBO Reports fourteen (2013): 759–764.
Glossary
- Le Chatelier's principle
- when a chemical organisation at equilibrium is disturbed, it returns to equilibrium by counteracting the disturbance
- position of equilibrium
- concentrations or fractional pressures of components of a reaction at equilibrium (commonly used to draw conditions before a disturbance)
- stress
- modify to a reaction's atmospheric condition that may cause a shift in the equilibrium
Source: https://chem.libretexts.org/Courses/University_of_British_Columbia/UBC_Introductory_Chemistry/11%3A_Chemical_Equilibrium/11.08%3A_Le_Chateliers_Principle-_How_a_System_at_Equilibrium_Responds_to_Disturbances
Post a Comment for "What Would Happen to the Equilibrium Expressed by the Reaction in Equation 11.8"